Consider the titration of 100.0 mL of 0.100 M HCN by 0.100 M KOH at 25°C. K for HCN 6.2×10-10, Part 1 Calculate the pH after 0.0 mL of KOH has been added. pH = Part 2 Calculate the pH after 50.0 mL of KOH has been added. pH = Part 3 Calculate the pH after 75.0 mL of KOH has been added. pH = Part 4 Calculate the pH at the equivalence point. pH = Part 5 Calculate the pH after 125 mL of KOH has been added. pH =
Consider the titration of 100.0 mL of 0.100 M HCN by 0.100 M KOH at 25°C. K for HCN 6.2×10-10, Part 1 Calculate the pH after 0.0 mL of KOH has been added. pH = Part 2 Calculate the pH after 50.0 mL of KOH has been added. pH = Part 3 Calculate the pH after 75.0 mL of KOH has been added. pH = Part 4 Calculate the pH at the equivalence point. pH = Part 5 Calculate the pH after 125 mL of KOH has been added. pH =
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter14: Acid- Base Equilibria
Section: Chapter Questions
Problem 105CWP: Consider the titration of 100.0 mL of 0.100 M HCN by 0.100 M KOH at 25C. (Ka for HCN = 6.2 1010.)...
Related questions
Question
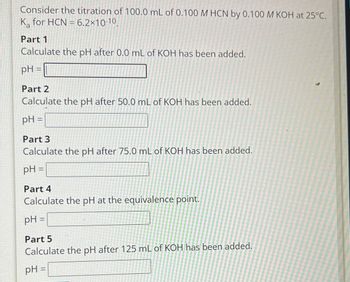
Transcribed Image Text:Consider the titration of 100.0 mL of 0.100 M HCN by 0.100 M KOH at 25°C.
K for HCN 6.2×10-10,
Part 1
Calculate the pH after 0.0 mL of KOH has been added.
pH =
Part 2
Calculate the pH after 50.0 mL of KOH has been added.
pH =
Part 3
Calculate the pH after 75.0 mL of KOH has been added.
pH =
Part 4
Calculate the pH at the equivalence point.
pH =
Part 5
Calculate the pH after 125 mL of KOH has been added.
pH =
AI-Generated Solution
Unlock instant AI solutions
Tap the button
to generate a solution
Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning